🟡 [FDA Debate Over Fluoride Supplements for Children]
While fluoride’s protective role in dental health is well-established, particularly through topical applications, the systemic ingestion model—especially in children—raises unresolved questions. The current FDA debate exposes a deeper structural tension: a product exempt from modern approval standards seeks renewed legitimacy under the banner of equity, despite low systemic efficacy and unclear long-term safety. The promotion of fluoride tablets reflects not only gaps in scientific consensus but also the persistence of symbolic public health interventions that may serve industrial supply chains more than patient needs. When effective topical alternatives exist, the systemic route becomes not a medical necessity, but a strategic and symbolic decision.
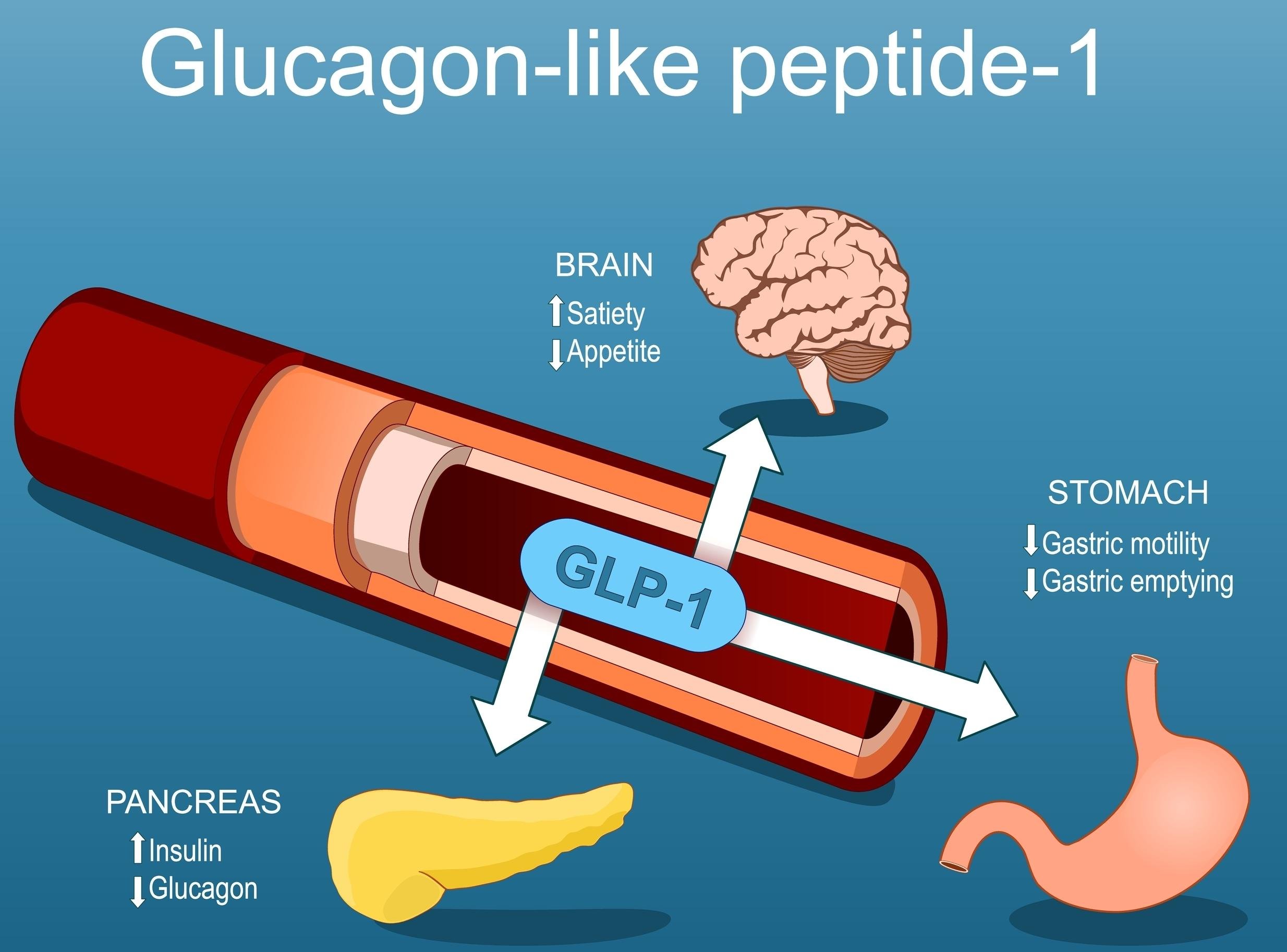
Evaluating the Risk of Lean Mass Reduction During GLP-1 Receptor Agonist Therapy
“While GLP-1 receptor agonists have revolutionized the treatment of metabolic disease, the emerging pattern of lean mass loss—accounting for up to 40% of total weight reduction in some trials—raises a clinically relevant paradox. Weight is lost, but so is structure. Without proactive intervention, this silent catabolism may compromise physical function, metabolic resilience, and quality of life. The therapeutic conversation must now evolve: from weight loss alone to functional preservation.”
🔎 FDA Rejects Brexpiprazole–Sertraline Combo for PTSD: Signal or Systemic Failure?
“Despite a mechanistically compelling rationale and one statistically positive Phase 3 trial, the FDA decisively rejected the brexpiprazole–sertraline combination for PTSD due to inconsistent efficacy and regulatory standards requiring replication. This case exposes the structural fragility of psychiatric drug development, where even a ~5-point improvement on CAPS-5 is insufficient without reproducibility. It also underscores a broader tension: between the urgent need for new PTSD treatments and the rigid evidentiary bar that neuropsychiatric indications must clear.”
The Hidden Patent Threat Undermining IntoCell’s Nexatecan PlatformA forensic patent analysis following ABL Bio’s sudden technology withdrawal
When ABL Bio abruptly returned the Nexatecan platform to IntoCell, few expected the root cause to lie beneath international waters — a Chinese submarine patent with striking structural similarities. This forensic comparison exposes the overlap between US20220047717A1 and WO2023088235A1, raising critical questions about freedom to operate, IP governance, and the hidden risks lurking in ADC development pipelines.
ProKidney Drives a Truly Advanced Cell Therapy for Diabetic CKD
Can an autologous cell therapy shift the course of diabetic kidney disease?
ProKidney’s REACT (Rilparencel) delivers a striking signal: a +4.6 mL/min/year improvement in eGFR slope in a Phase 2 study — one of the strongest functional preservation outcomes ever recorded in late-stage CKD.
But beneath the excitement lies a complex regulatory and methodological landscape. With no sham control, small sample size (n=49), and reliance on a surrogate endpoint, the data must be interpreted with precision.
This BBIU report dissects the trial design, FDA alignment, market reaction (NASDAQ: PROK +500%), and strategic outlook. It also frames the risks: regulatory uncertainty, lack of long-term durability data, and the challenge of scaling autologous manufacturing.
REACT is not a promise — it's a possibility. One that, if validated in Phase 3, could redefine what is therapeutically achievable in diabetic CKD.
Obicetrapib: Consistent Lipid-Lowering, Silent Systemic Risks
Obicetrapib: Beyond Lipids
The BROADWAY Phase 3 trial confirmed obicetrapib’s potent LDL-lowering efficacy (−32.6%) in high-risk cardiovascular patients already on maximal statins. But when efficacy is statistical and safety is probabilistic, the real question shifts:
What don’t we see—yet?
While clinical endpoints are reassuring, CETP inhibition raises mechanistic flags beyond plasma cholesterol:
Could rigidified macrophage membranes impair immune presentation?
Might hepatic cholesterol be silently accumulating?
Is hormonal balance vulnerable to long-term precursor depletion?
As obicetrapib moves from trial to population, its safety narrative must evolve from absence of harm to proactive detection of subtle biological shifts—before they manifest as clinical signals.
Mutation Without Smoke: How Urban Pollution Generates Genetic Signatures More Aggressive Than Tobacco
When pollution writes the genome.
In the largest study ever conducted on lung cancer in never-smokers, fine particulate pollution (PM2.5) was shown to trigger TP53 mutations—the same lethal genetic alterations caused by tobacco. Urban air doesn’t just irritate the lungs; it structurally reshapes the genome. As TP53-targeted immunotherapies emerge, we face a paradigm shift: pollution isn’t just a public health issue—it’s a mutagenic force with trillion-dollar implications for drug development, regulation, and justice. The data is no longer observational. It’s molecular.
What Happens to a Child During Economic Collapse? Part 3: Malnutrition, Tuberculosis, and the Future We Lose
What happens to a child during economic collapse?
They don’t go to the ICU.
They don’t file unemployment.
They disappear from the curve — quietly, biologically, structurally.
This report traces how economic breakdowns cause pediatric collapse through five interconnected pathways:
Chronic malnutrition
Neurocognitive regression
Tuberculosis reactivation
Institutional abandonment
Collapse of food infrastructure
We dissect how Isoniazid depletes vitamin B6, how MDR-TB bankrupts prevention, and how school kitchens can be repurposed as survival architecture — if properly shielded from contamination and bureaucracy.
This is not a humanitarian essay.
It is a structural field manual —
for when the collapse is no longer hypothetical.
What Happens to Health During an Economic Collapse?
In Part 2 of our series on structural health vulnerability, we dive into the silent erosion of respiratory stability during economic downturns.
High male smoking rates, rising female tobacco use, poor disease understanding, and unaffordable controller therapies converge into a pulmonary time bomb — especially in winter.
We show why asthma and COPD aren’t just clinical issues — they’re system indicators.
When they spike, it's already too late.
High-Precision Therapies vs. Collective Impact: The Case of Hemgenix and National Productivity
💡 What if curing disease isn't enough?
In 2025, the UK publicly funded Hemgenix — a $3.3M gene therapy for hemophilia B — offering life-changing benefits to a few dozen adults. But beneath the clinical triumph lies a structural question:
👉 Is this how we maximize collective return on public health investment?
This article explores the economic and ethical calculus behind breakthrough therapies, contrasting late-stage interventions with early, high-ROI strategies that preserve national productivity from childhood onward.
📊 From GDP modeling to risk-adjusted costs of lifelong transfusions, we analyze why innovation should align not just with individual outcomes, but with societal impact.
How a Clinical Trial Is Saved (or Ruined): Lessons from CTD Module 5
A critical breakdown of how clinical trial execution impacts the final CSR.
Based on direct experience, this piece walks through the CTD Module 5, SAE documentation, CRO collapse, CRA turnover, and why in-house oversight isn't optional.
CTD Module 4: The Cheapest Place to Prevent a $200M Loss
When companies talk about clinical development, most of the attention goes to the trial phases: Phase I safety, Phase II signals, Phase III failure or success.
But before all of that, there is a module—silent, technical, often overlooked—whose quality determines whether those millions should even be invested.
That module is CTD Module 4: Nonclinical Studies.
And its real value is not academic. It’s financial.
Prescribing in a Polypharmacy World: Why the Right Questions Matter More Than the Right Pills
Patients often don’t know the actual names, doses, or interactions of the medications they’re taking. Relying solely on verbal recall during consultations leads to incomplete, and sometimes misleading, information.
🚨 FDA Inspection After a Warning Letter: Consequences, Scenarios, and Strategic Response
When a pharmaceutical or biotech company receives a Warning Letter from the U.S. FDA, it's not just a regulatory alert. It marks the beginning of a critical period where decisions made in the next 30–90 days can determine whether the company recovers or descends into long-term damage.
Quality, SOPs, and Consequences: An Uncomfortable Truth in Pharmaceutical Manufacturing
Why fake SOPs lead to real disasters: unpacking the link between documentation, quality, and FDA enforcement.
The CTD as a Mirror: Operational Truths Behind Regulatory Success or Failure
When people talk about regulatory submissions and global expansion, they usually think of clinical efficacy, GCP compliance, or statistical robustness. But in practice, the real problems rarely occur where expected. Many product rejections are not caused by scientific failure, but by internal frictions within the companies themselves: delayed decisions, operational resistance, or misaligned documentation.

![🟡 [FDA Debate Over Fluoride Supplements for Children]](https://images.squarespace-cdn.com/content/v1/685a879d969073618e9775db/1753410105620-7LZN7YI3NNXHGHP6XTW1/istockphoto-1160757152-612x612.jpg)