Early Aspirin Withdrawal After PCI in Low-Risk Myocardial Infarction
In the post-PCI landscape shaped by new-generation drug-eluting stents, the antiplatelet decision is no longer a simple “DAPT by default” algorithm. The physician must evaluate a shifting pharmacodynamic hierarchy: aspirin provides limited incremental protection beyond the first month, while potent P2Y12 inhibition (ticagrelor or prasugrel) anchors the true anti-thrombotic effect. The modern question is not whether to withdraw aspirin, but when—and in which phenotype the balance between ischemic risk and bleeding liability makes early de-escalation both safe and structurally rational.
When Evidence Breaks: The Itvisma Approval, Novartis’ Faulty Reference, and the New SMA Power Hierarchy
Intrathecal delivery of onasemnogene abeparvovec requires a controlled lumbar puncture under sterile conditions, typically performed by a neurologist or anesthesiologist with experience in neuromuscular disorders. In older children and adolescents with SMA, the procedure enables direct vector exposure to spinal motor neurons while minimizing systemic AAV9 load. The clinical setting reflects the dual nature of the intervention: a one-time administration with the procedural complexity of a minor neurosurgical act and the transformative potential of gene replacement therapy.
The Structural Incompleteness of the modRNA Influenza Vaccine:Long-Term Safety Blindness, Inferential Fragility, and a Critical Immunogenicity Failure
The modRNA influenza platform shows statistical promise but remains structurally incomplete. Its biological half-life is unknown, its long-term immunologic consequences uncharacterized, and its efficacy estimate marked by severe inferential instability. Most critically, the vaccine fails to achieve noninferiority for influenza B, leaving a foundational gap in quadrivalent coverage. Until these structural deficiencies are resolved, the technology cannot be considered a reliable seasonal replacement.
Redefining Hemodynamic Strategy: How the EVERDAC Trial Resets the Standard of Care in Shock Management
Shock is not a blood-pressure problem — it is a perfusion failure. For decades, medicine responded to this failure with reflexive invasiveness, assuming that earlier arterial lines and tighter numerical control would save lives. The EVERDAC Trial dismantles that doctrine: critically ill patients in shock do just as well without early invasive catheterization, while avoiding the complications it introduces. The paradigm is shifting from procedure-first to physiology-first — restoring tissue oxygenation, guiding treatment with ultrasound, and reserving invasive tools only when the physiology demands them.
Beta-Blockers after Myocardial Infarction: When Physiologic Logic Outlives Clinical Reality
The 2025 New England Journal of Medicine pooled analysis marks the end of a 50-year reflex in cardiology. Among more than 17 800 patients who survived myocardial infarction with a preserved left-ventricular ejection fraction (≥ 50 %), beta-blocker therapy no longer conferred protection against death, recurrent infarction, or heart-failure hospitalization.
For decades, sympathetic blockade was viewed as an indispensable safeguard against electrical instability and sudden death. Yet in the modern reperfusion era—where nearly every patient receives stenting, dual antiplatelet therapy, statins, and renin–angiotensin inhibition—the old logic has dissolved. The drug’s physiologic rationale remains intact, but its clinical necessity has vanished.
Economically, this correction reshapes a USD 10 billion global class: up to 5 % of routine post-MI prescriptions will disappear, freeing hundreds of millions in low-value expenditure. What endures are the genuine indications—heart failure, arrhythmia, ischemic control—while medicine itself transitions from habitual protection to functional precision.
PSA Screening: Detection Without Prevention
The prostate-specific antigen (PSA) test was once hailed as a revolution in early cancer detection. Yet its promise of prevention has collapsed under the weight of biological ambiguity. PSA levels rise not only in malignancy but also in benign hyperplasia and inflammation, blurring the line between vigilance and harm. A normal prostate secretes small amounts of PSA into the bloodstream; as the gland enlarges or becomes cancerous, disruption of tissue architecture releases larger quantities. However, this increase is neither linear nor specific. Many men with elevated PSA never develop cancer, while some with aggressive disease show normal values.
The test thus measures disruption, not malignancy. It detects activity within the prostate but cannot distinguish its meaning. This diagnostic paradox—precision without discrimination—defines the core epistemic flaw of PSA screening: it finds disease where risk is low and often misses where risk is real.
FDA Removes the "Bridge Trial" Requirement for Biosimilars: Regulatory and Economic Implications
The Department of Health and Human Services (HHS) stands at the intersection of affordability, innovation, and public trust in the age of biosimilars.
Through the combined oversight of the FDA and the Centers for Medicare & Medicaid Services (CMS), HHS is now redefining how biologic equivalence is validated—not merely through clinical replication, but through structural, molecular, and economic coherence.
Biosimilars represent both a scientific triumph and a fiscal experiment: the ability to reproduce the therapeutic precision of originator biologics at lower cost, without eroding confidence or safety.
Under the 2025 guidance removing the “bridge trial” requirement, HHS moves toward a total-evidence framework, where analytical comparability and real-world pharmacovigilance replace redundant human trials.
This shift marks a new philosophy of governance: trust in molecular data, anchored by post-market integrity.
It is not deregulation, but reallocation—an evolution from procedural proof to structural truth.
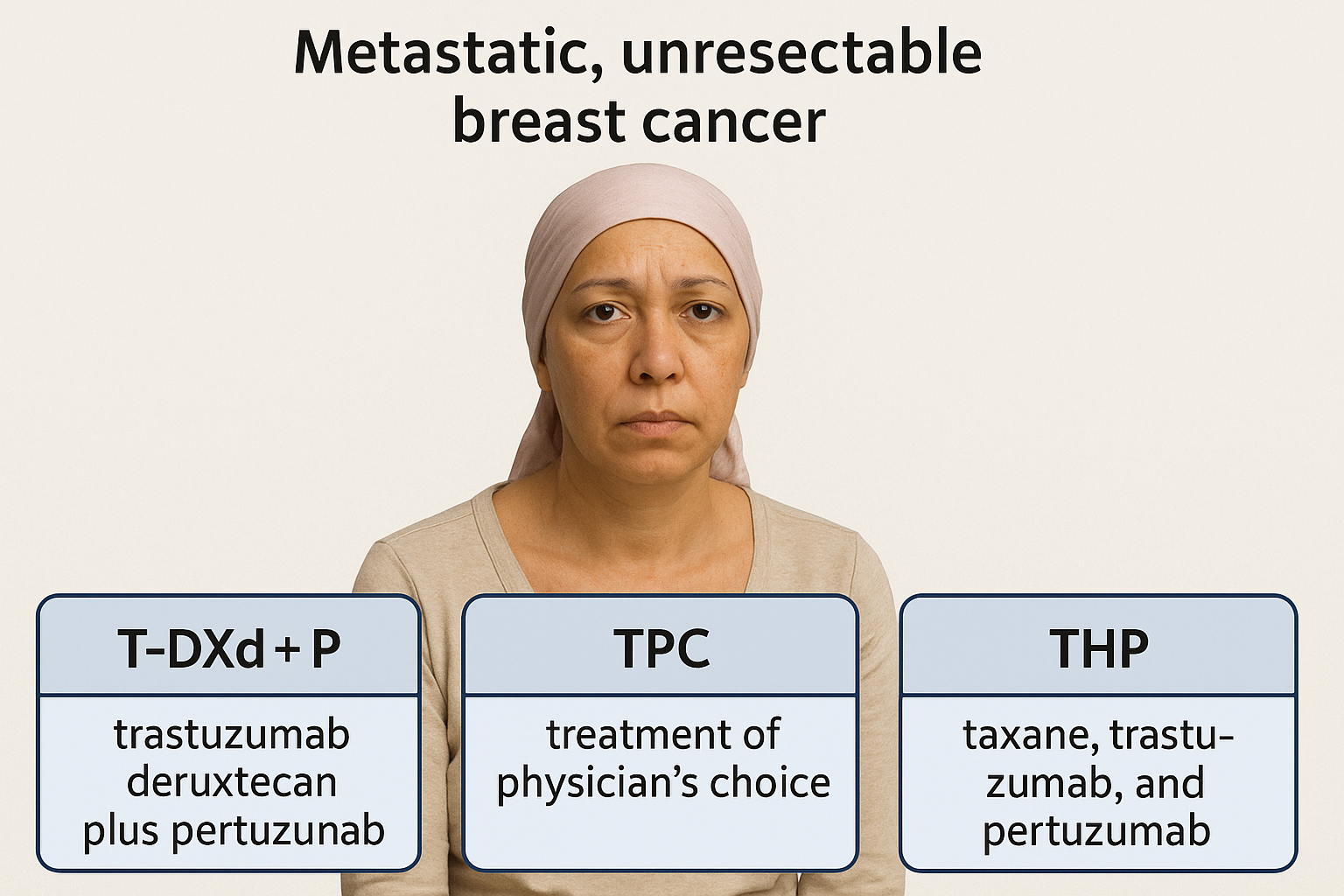
T-DXd + Pertuzumab Redefines First-Line HER2+ Therapy: Quantitative Supremacy, Structural Risk
In the quiet space between diagnosis and decision, a woman with metastatic, unresectable HER2-positive breast cancer confronts three divergent paths.
On one side, the long-familiar THP regimen — taxane, trastuzumab, and pertuzumab — the cornerstone of a decade’s treatment.
In front of her, the new architecture of antibody–drug conjugates: trastuzumab deruxtecan (T-DXd) plus pertuzumab, a design where cytotoxic precision replaces the blunt force of chemotherapy.
And to the third path, the unblinded promise of T-DXd monotherapy — still in shadow, awaiting full revelation.
Each arm is more than a protocol. It is a symbol of how oncology now defines hope:
by balancing efficacy against toxicity, progress against opacity, and survival against the integrity of truth revealed in stages.
Annex 1 — Pharmacological Composition and Adverse-Event Profiles
Annex 2 — Early-Phase Clinical Evidence of Trastuzumab Deruxtecan (Phase 1 and 2)
Annex 3 — Pharmacoeconomic Impact and Health-System Integration Forecast
Annex 4 — Financial and Strategic Market Impact (AstraZeneca & Daiichi Sankyo)
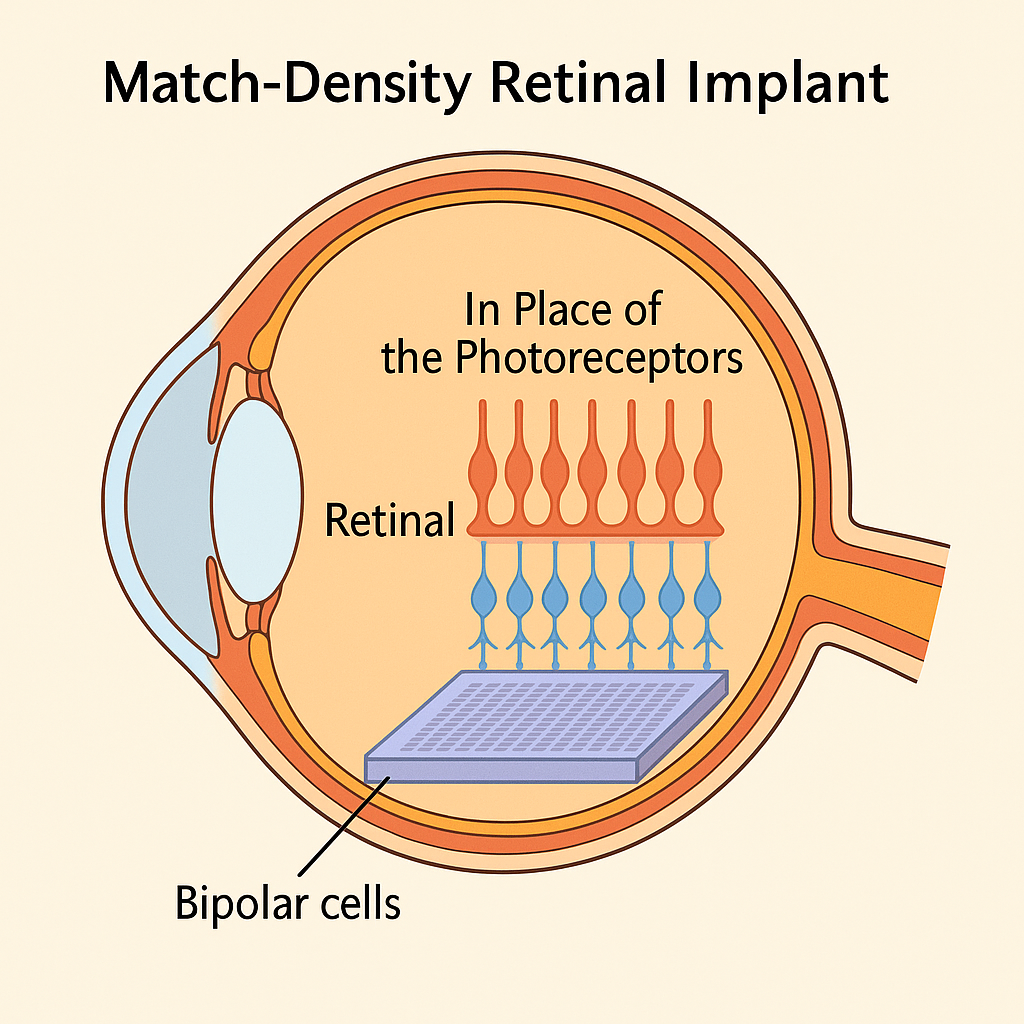
Restoration of Central Vision With the PRIMA System
The PRIMAvera trial, published in NEJM (2025), demonstrates for the first time that a subretinal photovoltaic implant can restore central vision in patients with advanced dry AMD. At 12 months, 81% of evaluable patients achieved clinically meaningful visual gains, including word and number recognition. Yet the success rate falls closer to 65–70% on an intention-to-treat basis, revealing a structural gap between published efficacy and enrolled reality.
The deeper lesson lies beyond today’s results: the human retina has ~25–30,000 bipolar neurons per mm², while the PRIMA chip offers only ~95 electrodes/mm². Semiconductor technology could easily deliver millions of pixels, but overcrowding would cause thermal and electrochemical damage without adding useful vision. The future is not about raw density but about matching electrode arrays to biological capacity. By aligning implant design with natural limits—and leveraging blinking, pupillary reflex, and cortical plasticity—retinal prosthetics may finally cross from experimental restoration to functional daily use.
Annex Title: Biological Match Design for Subretinal Nano-Fabrication
Common Diseases, Hidden Rarities: The 3% Logic That Challenges the 97%
A recent NEJM study (Rahimov et al., 2025) revealed that up to 3% of patients diagnosed with common diseases such as multiple sclerosis, inflammatory bowel disease, or atopic dermatitis in fact harbor rare monogenic disorders. These hidden cases distort clinical trial outcomes, dilute efficacy signals, and misdirect patient care. The paradox is evident: to capture a small minority, one must consider testing the entire majority. Yet the rational solution is stratification — sequencing reserved for non-responders or applied during biopsy-confirmed disease. This approach reduces costs by two-thirds, triples cost-effectiveness, and maintains clinical integrity. In clinical trials, genetic screening as an inclusion criterion yields smaller but purer cohorts, while regulators will demand strict SAE oversight and 3–5 years of enhanced pharmacovigilance post-launch. Precision becomes sustainable only when anchored to feasibility.
Annex 1 — The Diagnostic Journey
Annex 2 — Health Economics of Sequencing
Annex 3 — Genetic Screening as an Inclusion Criterion
Elecsys pTau181: FDA Approval, Clinical Promise or Structural Market Expansion?
On October 13, 2025, the FDA approved Roche and Eli Lilly’s Elecsys pTau181 blood test, marking the second regulatory clearance of a blood-based assay for Alzheimer’s disease, following Fujirebio’s earlier approval in May. The Elecsys test, designed for adults over 55 with cognitive complaints, measures phosphorylated tau181 in plasma and is intended as a rule-out tool rather than a confirmatory diagnostic.
Clinical data show a negative predictive value (NPV) of 97.9%, allowing physicians to reliably exclude Alzheimer’s pathology in primary care. However, specificity remains limited at ~70%, restricting its standalone diagnostic utility. While the test does not modify therapeutic decisions—treatment initiation still requires PET or CSF confirmation—it creates a scalable entry point into everyday practice.
From a structural perspective, Elecsys embodies the tension between clinical necessity and industrial expansion: marketed as democratization of diagnosis, yet strategically positioned to embed Roche’s Elecsys platform into routine workflows, potentially inflating healthcare costs through repeat testing and downstream imaging.
Annex 1 — Alzheimer’s Disease
Annex 2 — Technical Basis of the Elecsys pTau181 Blood Test
Annex 3 — Related Patent Landscape with Expiry Dates
Annex 4 — Potential Market Size and Estimated Revenue
Sacituzumab Tirumotecan in EGFR-Resistant NSCLC: A Two-Layer Integrity Analysis
The clinical development of sacituzumab tirumotecan (SKB264) illustrates a profound contradiction between scientific promise and ethical responsibility. While early-phase studies generated signals of efficacy in EGFR-mutant NSCLC, the absence of prospective TROP2 confirmation represents a fundamental breach of clinical and research standards. Biopsy-based molecular profiling is routine worldwide in NSCLC; to proceed without confirming the presence of the drug’s target exposed patients to risk without a reasonable expectation of benefit.
Moreover, the inflated enrollment in phase 1/2 (over 1,400 patients across >100 centers) contrasted with the China-only pivotal phase 3 (~356–480 patients) creates a transparency gap. The large discrepancy between subjects enrolled and those reported in publications leaves open the possibility of selective reporting and statistical manipulation. Together, these design flaws compromise both the ethical and epistemic integrity of the SKB264 program.
BBIU concludes that this is not simply a matter of methodological weakness but a systemic ethical lapse: patient rights to informed consent, standard of care, protection from harm, scientific integrity, and equal treatment have been compromised. Regulatory shortcuts—accepted under FDA IND flexibility in early phases and then restricted to NMPA approval in China—magnify the credibility gap.
Annex 1 — BBIU Regulatory Due-Diligence Checklist for ADC Trials
Annex 2 — Ethical Violations and Patient Rights Breaches in SKB264 Trials
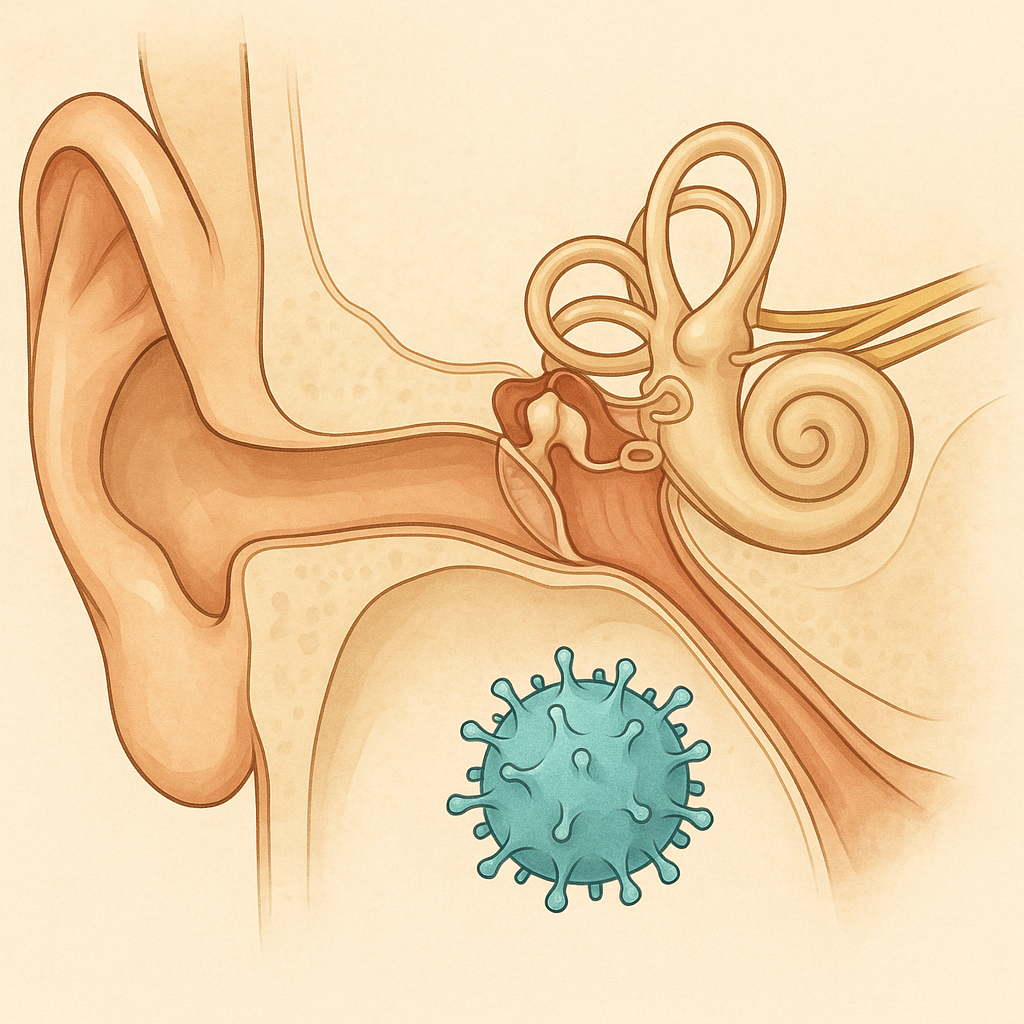
Restoring Hearing Through Otoferlin Gene Therapy: Structural Implications of DB-OTO Trial
The DB-OTO trial (NEJM, 2024) shows first evidence that congenital deafness from OTOF mutations can be partially reversed. Children treated with a dual-AAV cochlear injection recovered auditory responses within weeks. This is a milestone in sensory gene therapy, but early framing overstated efficacy: the protocol’s primary endpoint is safety, not hearing restoration. With <15 patients so far, long-term risks and durability remain unknown.
Annex 1 — Otoferlin Biology and Clinical Translation
Annex 2 — Dual AAV and Immune Risks
CDC’s Retreat from Universal COVID Vaccination: Structural Implications for Public Health Trust
On October 7, 2025, the CDC made a historic change to its immunization schedule: COVID-19 vaccines are no longer universally recommended for all Americans six months and older. Instead, vaccination is now an individual decision, to be made in consultation with a physician.
At the same time, the CDC adjusted pediatric guidelines: toddlers will now receive separate MMR and varicella shots rather than the combined MMRV, after studies showed a slightly higher risk of febrile seizures with the combo dose.
The shift reflects a broader recalibration of U.S. public health policy—from blanket mandates toward individualized consent—amid political reshaping of the CDC’s advisory committee and declining public confidence in universal vaccine strategies.
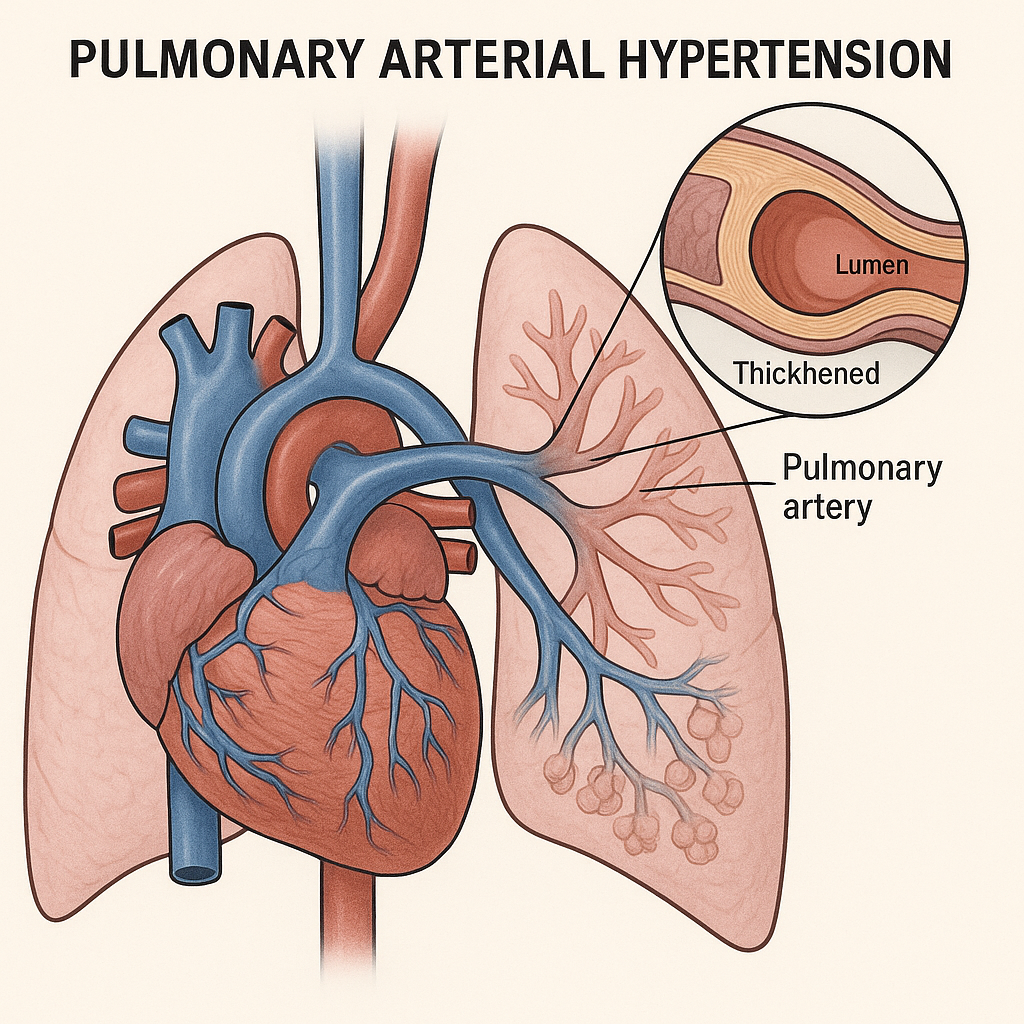
Sotatercept in Early Pulmonary Arterial Hypertension: Structural Reassessment of Therapeutic Onset
What’s new (NEJM, Sept 30, 2025; HYPERION, NCT04811092).
First Phase 3 use of sotatercept within ≤12 months of PAH diagnosis on top of dual/triple therapy. Primary endpoint met: clinical worsening 10.6% vs 36.9%; HR 0.24 (95% CI 0.14–0.41; P<0.001). Fewer hospitalizations (1.9% vs 8.8%) and exercise deterioration (5.0% vs 28.8%); mortality neutral at 1 year. Trial stopped early for loss of equipoise.
Why it matters.
Sotatercept shifts from late “rescue” to early disease-modifying anchor, likely pressuring guidelines, payers, and treatment algorithms.
Annex 1 (Pathophysiology) — One-page gist
Annex 2 (Sotatercept Pharmacology) — Essentials
Annex 3 (Pharmacoeconomics) — Bottom line
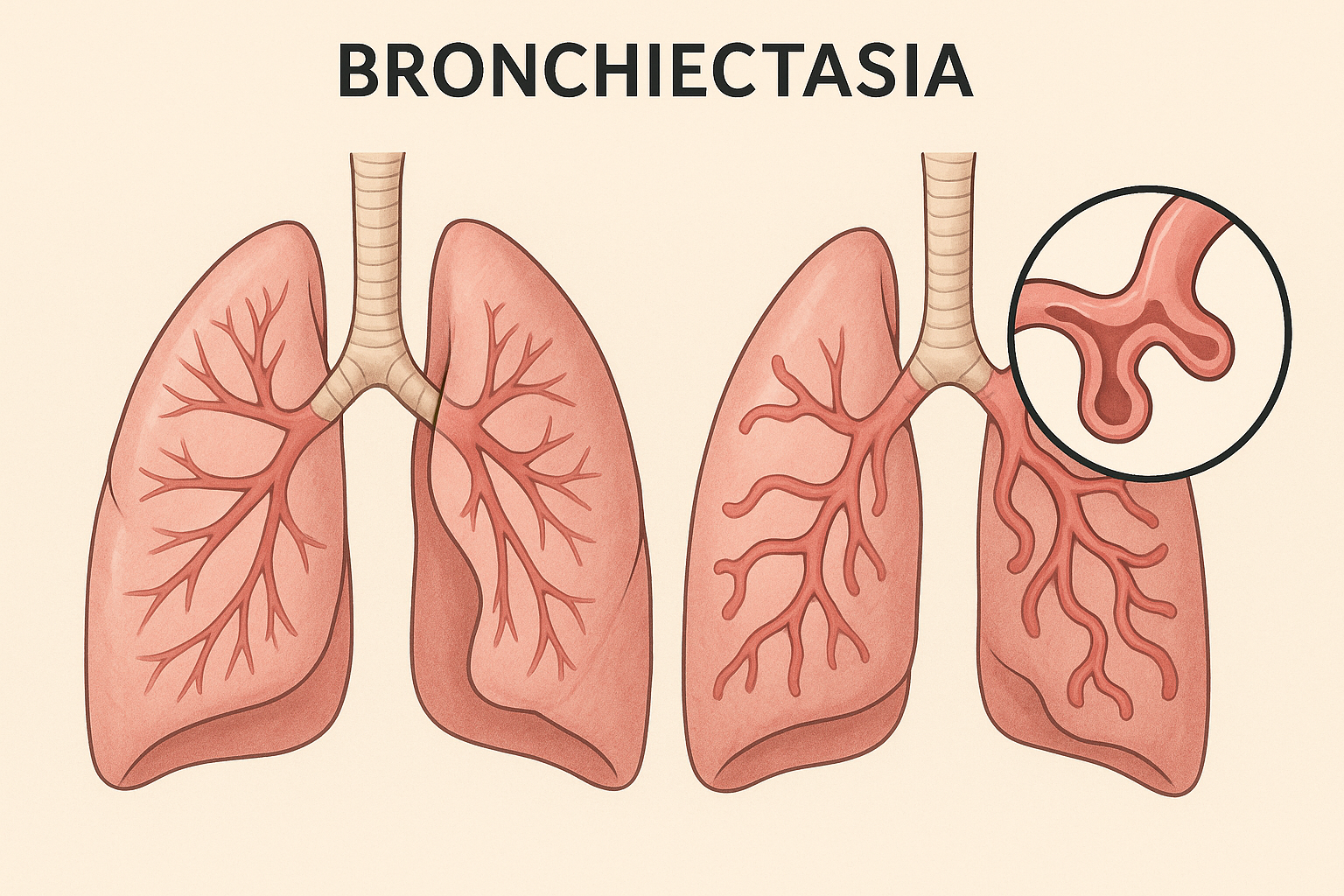
NEJM Trial on Mucoactive Therapies in Bronchiectasis: Structural Reassessment of Clinical Value
Trial at a glance (CLEAR, NEJMoa2510095).
Multicenter UK, 2×2 factorial, n=288, 52-week follow-up. Interventions: 6% hypertonic saline (inhaled) and carbocisteine (oral) vs standard care.
Primary outcome: annualized exacerbations — no significant reduction (adj. Δ: saline −0.25; carbocisteine −0.04).
Secondaries: no consistent benefit in QoL, lung function, or time-to-exacerbation. Safety acceptable.
Implication.
Guideline habit of universal mucoactive use in non-CF bronchiectasis is structurally weakened; shift toward selective, symptomatic use rather than routine prescription.
Annex Snapshot — Technical Evidence Base (Key Points)
Inhaled Heparin: An Old Anticoagulant Emerging as a New Weapon Against Severe Respiratory Infections
What’s new. A multinational meta-trial (eClinicalMedicine, 2025) found nebulized unfractionated heparin cut the combined risk of intubation or death by ~50% in hospitalized COVID-19 patients versus standard care—reigniting interest in a cheap, stockpiled therapy.
Why it matters. Current tools are fragmented (early antivirals, broad steroids, pricey biologics). Inhaled heparin concentrates action in the lungs with minimal systemic exposure, making it attractive for low- and middle-income settings and surge waves.
How it works (multi-pathway).
Anticoagulant: counters pulmonary microthrombosis.
Anti-inflammatory & endothelial protection: stabilizes the alveolar–capillary barrier.
Antiviral decoy: can bind spike proteins and impede entry.
Safety/unknowns. Generally reassuring with inhalation (lower systemic absorption → lower HIT risk), but dose, schedule, and bleeding surveillance need Phase III confirmation.
Mini-Annex (snapshots).
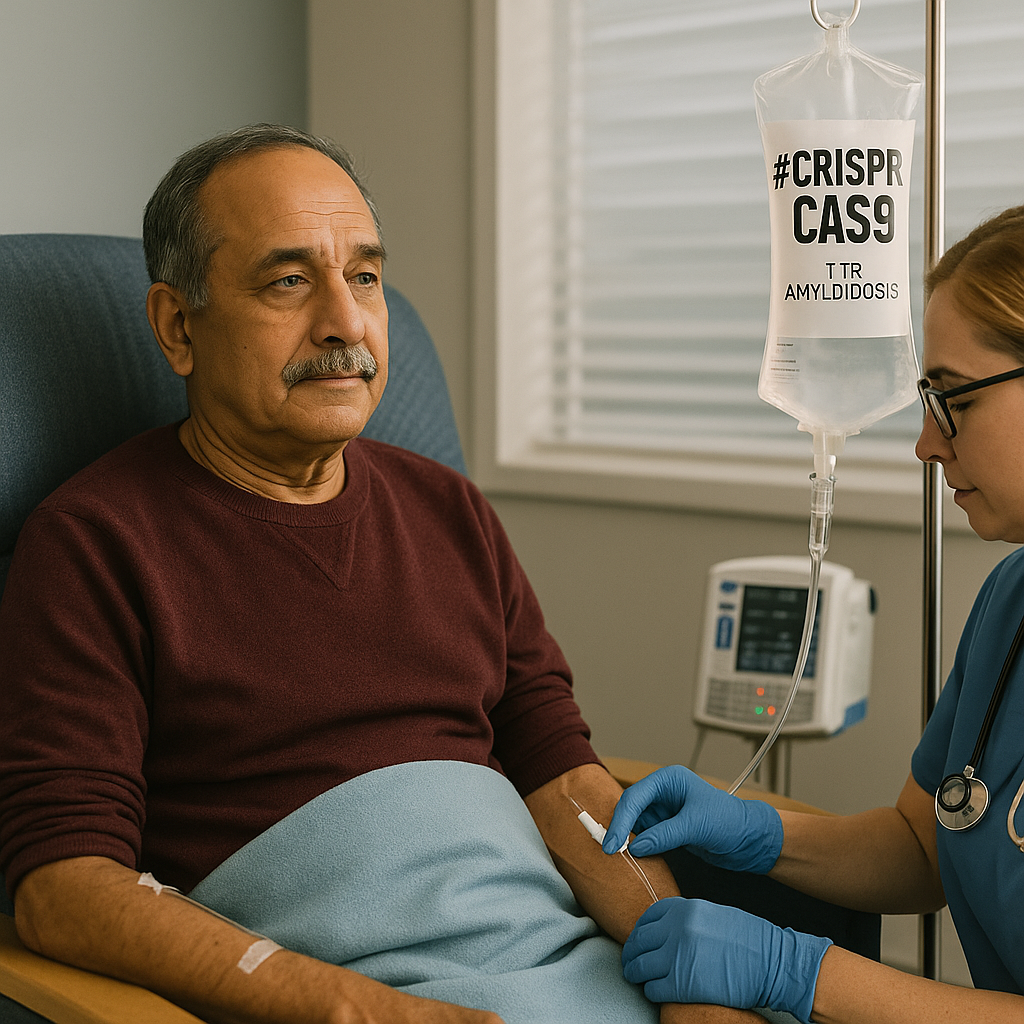
Nexiguran Ziclumeran Gene Editing in Hereditary ATTR with Polyneuropathy
The NEJM trial of nexiguran ziclumeran (NTLA-2001) marks the first demonstration of systemic in vivo CRISPR editing in humans. By targeting hepatic TTR production, patients with hereditary transthyretin amyloidosis achieved >90% median serum TTR knockdown and stabilization of neuropathy scores. While safety signals were generally favorable, the study’s early-phase design leaves key uncertainties: attribution of serious adverse events, long-term off-target effects, and the consequences of lifelong TTR silencing. Beyond ATTRv-PN, this trial symbolizes a turning point—patients act simultaneously as treatment recipients and pioneers in proving the feasibility of permanent gene editing for chronic disease.
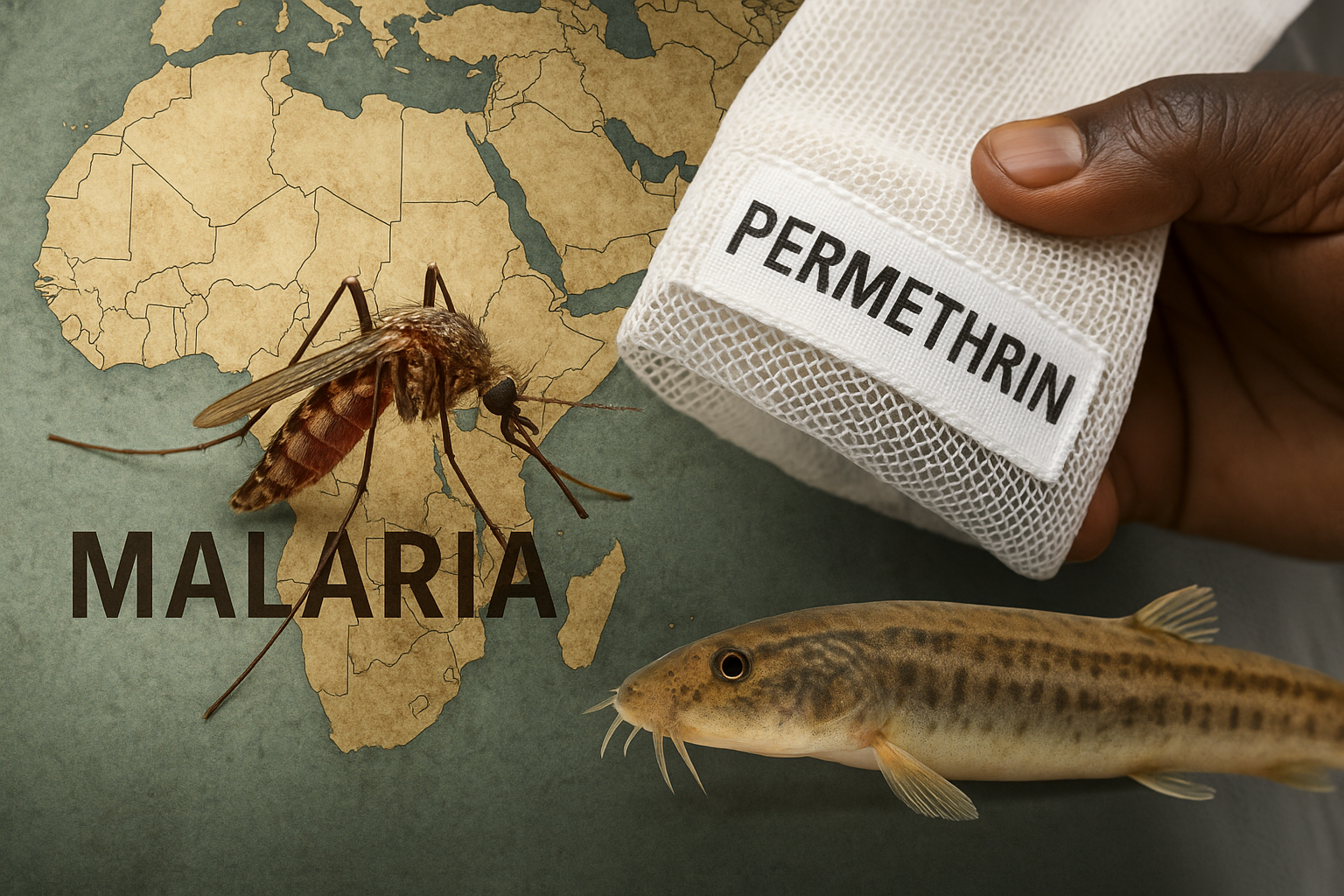
Permethrin-Treated Baby Wraps: An Experimental Shield Against Malaria
The NEJM trial on permethrin-treated baby wraps reveals both the promise and the peril of chemical vector control in Africa. While the wraps reduced malaria incidence among infants, their long-term use risks ecological toxicity and accelerated resistance in Anopheles mosquitoes. In contrast, the dojo loach (Misgurnus anguillicaudatus) offers a bioeconomic pathway: natural larval predation, improved community nutrition, and sustainable resilience. The future of malaria control will not be decided by chemistry alone, but by whether we choose dependency or ecological sovereignty.
FDA Blocks Biogen’s High-Dose Spinraza in the U.S.: A Regulatory Setback Driven by Manufacturing Controls
On September 23, 2025, the FDA issued a Complete Response Letter (CRL) rejecting Biogen’s high-dose Spinraza submission, not due to safety or efficacy, but because of deficiencies in Module 3 (CMC). This dossier, the cornerstone of manufacturing integrity, failed to convince regulators that the higher-dose formulation could be produced consistently and reliably at scale. The rejection highlights a structural miscalculation: Biogen’s decision to prioritize speed under commercial pressure, while leaving critical documentation incomplete. It is a reminder that in today’s regulatory landscape, the ultimate gatekeeper of therapeutic progress is not clinical promise, but manufacturing rigor.